|
3/17/2024 0 Comments Bohr atomic models 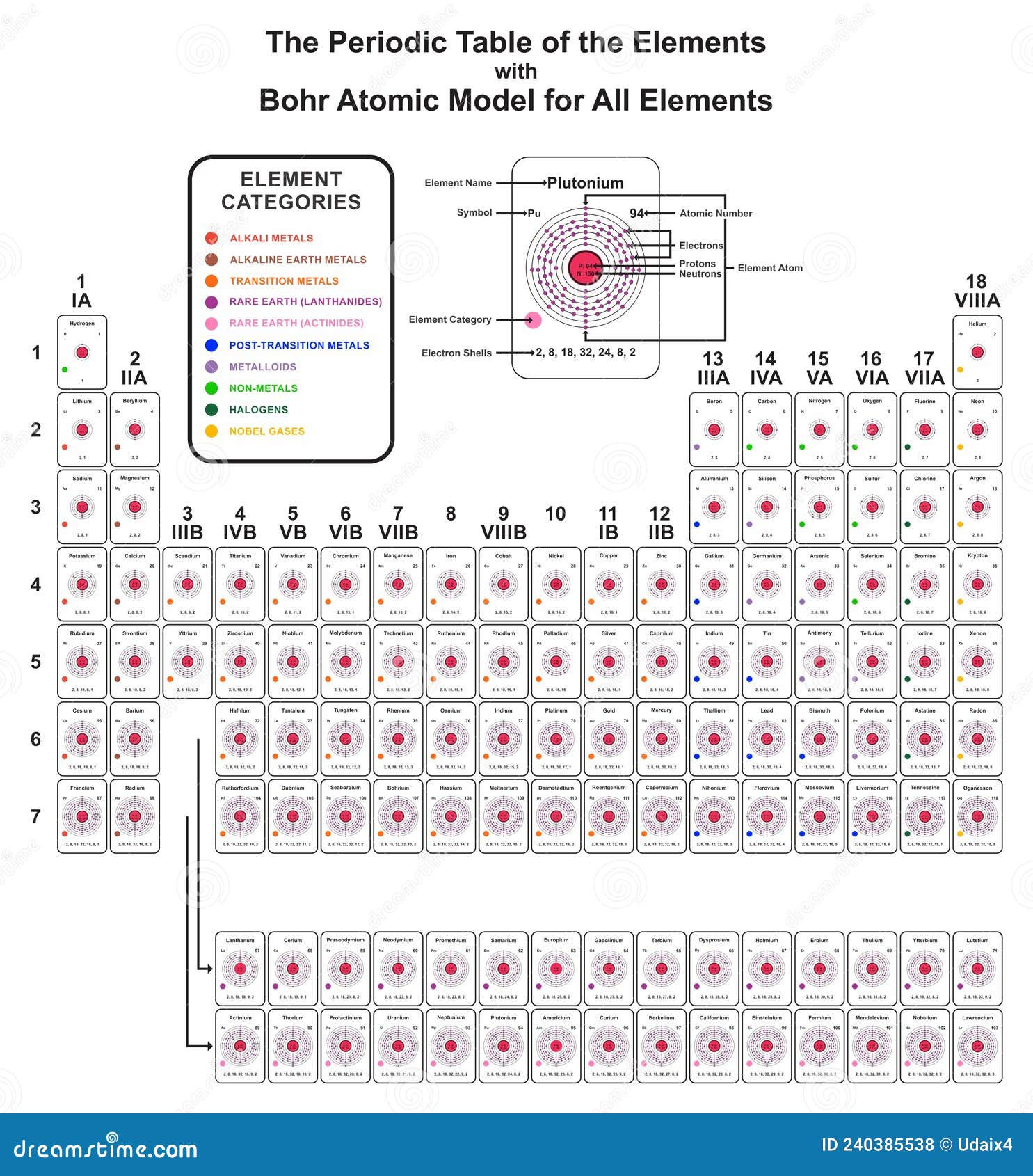
Through the synthesis of these models, physicists continue to delve deeper into the enigmatic realm of quantum physics, unraveling the mysteries that govern the fundamental building blocks of our universe.Isn't to do with the fact that the velocity is not quite 0? if you know it is exactly 0 then the uncertainty in the position is infinite as well (momentum is a function of velocity, so delta P = 0 -> delta V = 0 -> delta X = inf) therefore it has an equal probability of being anywhere. The evolution from Bohr’s simplistic yet groundbreaking model to the sophisticated and probabilistic Quantum Mechanical Model stands as a testament to the relentless pursuit of knowledge and the ever-evolving nature of scientific understanding. Both models, though distinct in their approaches, contributed significantly to the progression of atomic theory, shaping our understanding of the microscopic world. While Bohr’s model laid the groundwork for understanding atomic structure, the Quantum Mechanical Model surpassed it, offering a more comprehensive and accurate depiction of the behavior of subatomic particles. Consideration of Wave Properties: The Quantum Model accounts for the wave-like nature of particles, a concept absent in Bohr’s model.Mathematical Formalism: The Bohr Model uses classical mechanics and simple equations, whereas the Quantum Model relies on complex mathematical equations of quantum mechanics.Treatment of Orbits: Bohr’s model assumes fixed orbits, while the Quantum Model describes the electron’s position as a probability distribution.Predictive Power: Both models successfully predict atomic spectra and certain behaviors of electrons.ĭifferences Between the Bohr and Quantum Models.Quantized Energy Levels: Both models recognize the existence of discrete energy levels in an atom.Similarities Between the Bohr and Quantum Models Uncertainty Principle: Introduced by Werner Heisenberg, it asserts that it’s impossible to simultaneously know the exact position and momentum of an electron.Probability Clouds: Rather than fixed orbits, the model presents the electron’s probable location within an electron cloud, characterized by regions where electrons are likely to be found.Wave-Particle Duality: Electrons exhibit both particle-like and wave-like behaviors, described by the wave function in Schrödinger’s equation. 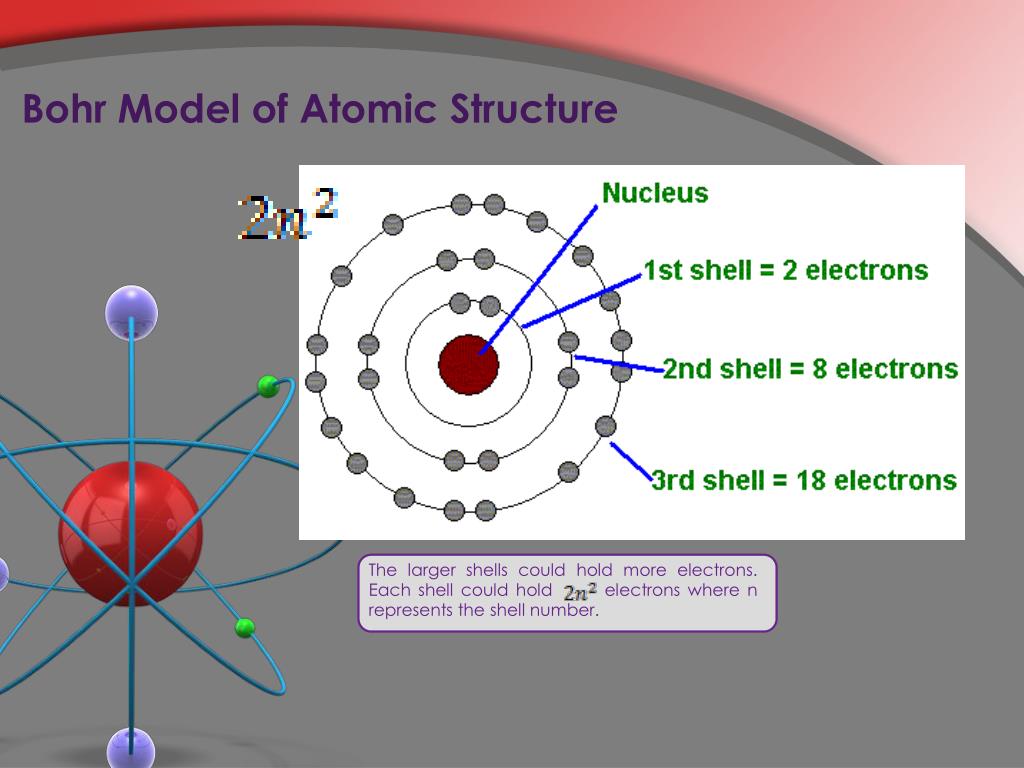
This model, based on the principles of quantum mechanics, introduced profound changes to our understanding of atomic structure: The Quantum Mechanical Model emerged as a breakthrough in the 1920s, building upon Bohr’s ideas and incorporating quantum theory.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
